One of the big problems when it comes to treating cancer using drugs is that these drugs flood the patient’s body and cause detrimental side effects when they reach areas other than the malignant tumour. Cancer cells are derived from our own healthy cells – they’re hard to target specifically without also hitting our healthy cells. A lot of research goes into trying to get around this problem. I came across a particularly interesting study that I thought I’d share here.
Earlier this year Sofie Snipstad et al. published a paper in Ultrasound in Medicine & Biology titled “Ultrasound improves the delivery and therapeutic effect of nanoparticle-stabilized microbubbles in breast cancer xenografts”. This paper was particularly cool because of the rationale behind it. Nanoparticle delivery of drugs is something scientists have been working on for a little while. The premise is that you take your drug and you wrap it up inside a small protective bubble allowing the drug to travel to a specific site before it is released. Due to a quirk of cancer biology, this is particularly great as a cancer therapy. When tumours grow, they start to form their very own blood supply – only the blood vessels that they grow are more leaky than normal blood vessels. They allow slightly larger molecules to pass through from the blood vessel and into the surrounding tumour. This means we can use nanoparticles to deliver cancer drugs specifically into tumour sites by allowing the particles to travel through these leaky blood vessels. But then we hit another problem – if the tumour blood supply doesn’t reach the very depths of the tumour then the particles are too big to get all the way through. You can treat the edges but not the very centre of the tumour. So, this paper worked on a special combination – they took a bunch of nanoparticles containing chemotherapy and they bundled them up together into microbubbles that can travel around the blood system easily and safely until they reach the tumour site. Then, the researchers used a focused shot of ultrasound to break up the bubbles and release the nanoparticles. This also served to allow gentle tissue massage by the ultrasound to allow the nanoparticles to distribute further throughout the tumour. Once in the tumour, the cancer cells start to take up the nanoparticles and inside the cell the drug is released and can kill the cancer cell from the inside.
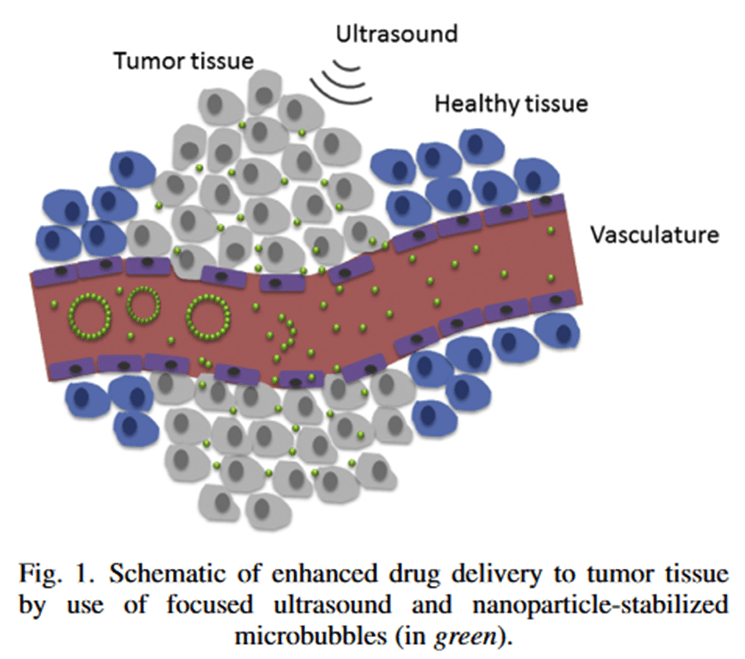 Image: figure from the paper
Image: figure from the paper
Any microbubbles that weren’t in the tumour site and therefore not exposed to the focused ultrasound could be easily cleared from the body without releasing the drug which means the only cells targeted by the therapy are the cancer cells. This means we can hit the cancer cells with a higher, more toxic dose because the healthy cells are not going to be hit with the same dose.
The researchers in this paper were testing the optimal way of doing this in mice suffering with triple negative breast cancer – one of the more aggressive forms of the disease – with positive results. The mouse tumours took up the drug 2.3x better and there was no tissue damage identified. All of the tumours either regressed or else the mice went into complete remission. The authors described this as a “promising proof-of-concept study”.